Batteries

Key Points
- Primary batteries are used once while secondary batteries can be used again and again
- There are clear patterns in battery energy that you can use to sketch out a power design
- Lithium-based batteries dominate the industry now
Introduction
All spacecraft use batteries. They’re used, for instance, at the beginning of a mission to power everything until solar panels and nuclear devices are started. In this article, we’ll talk about the different types and then talk a bit about how to size your battery system.
Battery Types
There are two broad categories: Primary batteries and Secondary batteries.
Primary Batteries
Primary batteries can only be used once. The AA batteries you buy at the store, for example, are “primary batteries”. After they’re drained, they’re essentially dead weight.
On spacecraft, they’re most often used at the beginning of the mission to help deploy solar panels, power mechanisms, and boot the computer. Because this is such a critical part of the mission, primary batteries typically:
- Have the highest chance of working without failure
- Are stable with long shelf lives
- Operate over a wider range of temperatures than secondary batteries
- Have mostly flat discharge curves so you get full performance throughout their life
Secondary Batteries
These are rechargeable batteries. On spacecraft, they’re often charged by solar panels and sometimes by nuclear generators. Over time, and especially with improvements in Lithium-ion technology, secondary cells have become increasingly trustworthy, to the point that some spacecraft don’t use primary batteries.
Battery Chemistries
There are three major types of chemistries you find in space batteries: Nickel, Lithium, and Silver. Nickel used to fly on most spacecraft because they lasted the longest, handled extreme heat and cold better, and (usually) failed gracefully.
Nowadays, though, the energy density of Lithium batteries is so attractive that they’ve mostly replaced Nickel, even though Lithium has more engineering challenges. For instance, Lithium has an explosion risk but battery vendors have done a good job engineering layers of safety features. Even the ISS, which goes to great lengths to protect its crew, uses them now.
Nickel-based
Nickel chemistries found in space systems include Nickel Metal Hydride (NiMH), Nickel-Cadmium (NiCd, pronounced Nigh-Cad), and Nickel-Hydrogen (Ni-H2).
Nickel-Cadmium batteries used to be everywhere. They’re stable, can be charged/discharged many thousands of times, their voltage decay curve is predictable, they can withstand large temperature ranges, and they’re safe to handle. However, their specific energy is low at around 55 W-hrs/kg (vendor numbers vary).
Some spacecraft used Nickel Metal Hydride batteries because their specific energy is around 65 W-hrs/kg. When every kilogram counts, that extra energy is appealing. However, NiMH can’t be cycled as many times as NiCd and they usually can’t sustain the high Amp draws that NiCd can. And they can’t get quite as cold either: NiCd can get to -40 ℃ whereas NiMH tends to hit its floor around -20 ℃. The challenges (especially the cycle limits) are hard enough that the extra energy density often wasn’t felt to be worth it.
Starting around the 1990s, Nickel-Hydrogen solutions started replacing NiCd solutions. Its energy density is about 70 W-hrs/kg, so about 30% more energy per kilogram than NiCd. It also can handle overcharging and discharging better. They can last for 50,000+ cycles and have been proven to power satellites for over 15 years. The International Space Station, for instance, used this type of battery for decades. If Lithium-based batteries hadn’t come along, this would probably still be what most spacecraft used.
Lithium-based
Space-rated, Lithium-based battery cells can have energy densities of 120 to 140 W-hrs/kg, which can be twice what a Nickel-based cell offers. That’s the kind of big leap that makes engineers smile in their sleep at night so it’s no surprise that Lithium now dominates the space battery market. There’s good news, too, in that Lithium is more shelf-stable. Nickel batteries can lose 20% of their energy every month they sit around while Lithium loses roughly 10%. This is one reason why many satellites have replaced their primary batteries with only secondary Lithium options.
It’s not all good news, though. Lithium can explode if charged the wrong way so extra engineering goes into ensuring its safe use. And Lithium doesn’t like to go below 0 ℃ (recall that Nickel can get down to -60 ℃), so extra engineering goes into keeping them warm. And Lithium cells can’t discharge at the same high amp rates as Nickel either so if you need high amps then you have to engineer the battery pack. And Lithium batteries tend to be more expensive than Nickel, too.
Despite all the challenges, it’s difficult to walk away from twice the energy storage per kg. And companies that specialize in space batteries have done a great job engineering solutions to the challenges. Although Nickel will continue to play a niche role in space, Lithium is expected to dominate in the foreseeable future.
Silver-based
Silver-Zinc and Silver-Oxide batteries are very high energy density options: they can have values over 200 W-hrs/kg which is higher even than Lithium solutions. Unfortunately, Silver-Zinc cells don’t do well with recharging because of dendritic growth. And Silver-Oxide can’t be recharged at all. However, if you just need to use them once then they’re a great option, which is why they show up on spacecraft as primary batteries.
Even the best primary battery becomes dead weight once it’s drained, though, so most spacecraft engineers still prefer reusable Lithium cells. Exceptions are on critical satellites such as high-end communications platforms which can afford the mass in exchange for extra certainty that they’ll have initial power. They’ll use both primary batteries and fully charged secondary batteries as a backup.
Series vs. Parallel Quick Notes
Batteries can be configured in series and/or in parallel.
Series: This is when you put one battery cell after another like railroad cars on a track. When you add them in series, you add the voltage. If each cell gives you 1V then putting ten cells in series gives you 10V.
Parallel: This is when you put them side-by-side like horses pulling a carriage. When you add them in parallel, you add their Amp-hours. So if a cell would last one hour for a given load, having two cells in parallel would make your system last two hours.
There are a few things to watch out for when combining cells:
- You’ve got to carefully match cell capacities and voltage profiles or the weakest one can drag down performance. Battery manufacturers have processes to ensure cells are well-matched.
- For critical systems, you need equipment to monitor the behavior of each cell and be able to shut it down without affecting the rest of the battery. CubeSats probably won’t have this feature but large spacecraft usually do.
- Especially for Lithium batteries, you need to make sure you charge each cell evenly. You don’t want to overcharge one misbehaving cell because you’re not monitoring it.
Plots
The following plots were created by collecting battery data from online resources. I’ve generalized them so we can talk about trends.
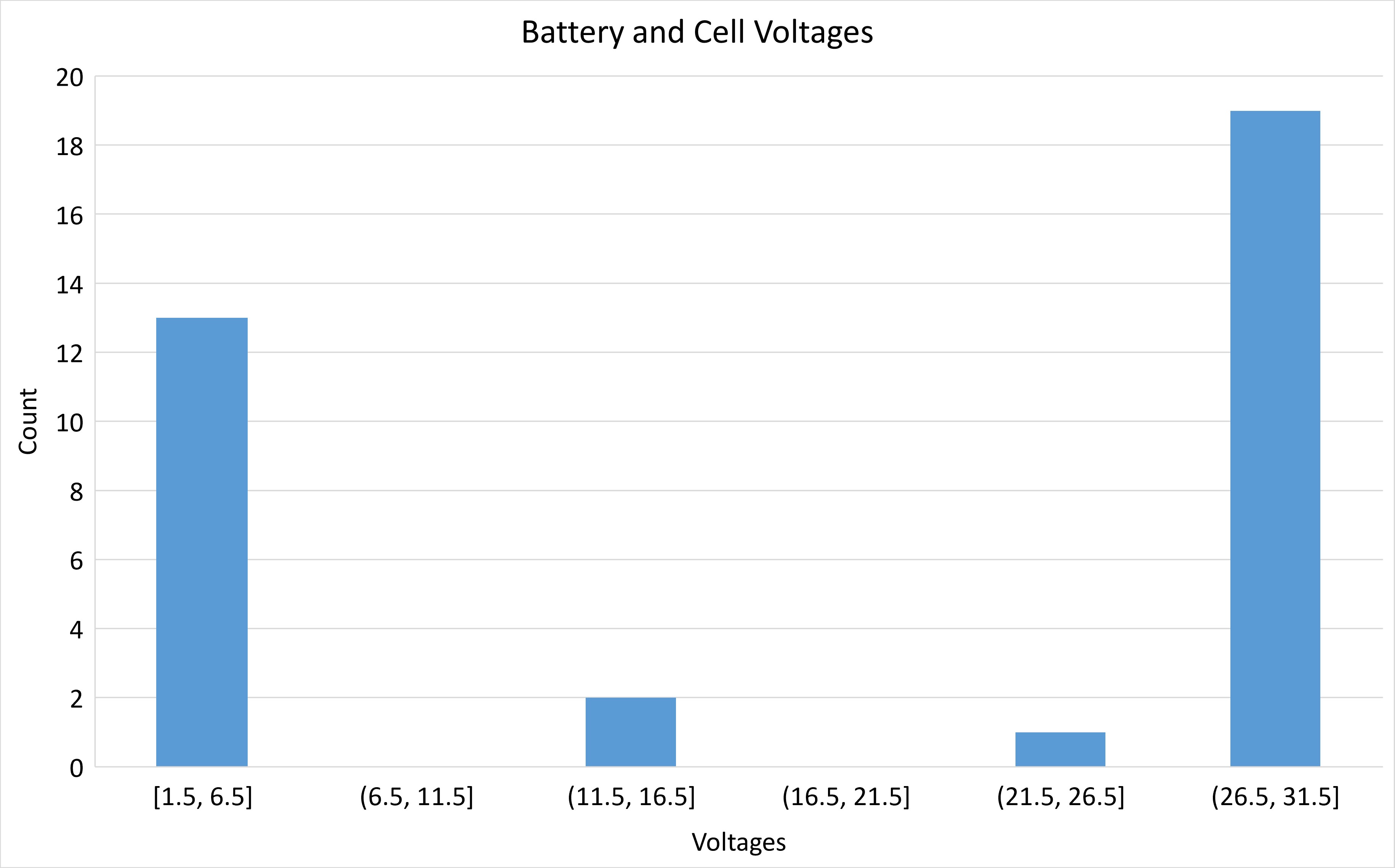
Voltage Categories
The first chart shows that battery vendors tend to sell things in a few categories. You can see that they target around 3.3 V (like what small electronic boards use) or around 28 V, which is what most large spacecraft use, similar to airplanes.
Energy vs. Mass
This plot has all sorts of battery types in it including Lithium-ion, Lithiated Nickel Cobalt Aluminum Oxide, and Silver Zinc. The plot shows that regardless of what type of battery you choose, the energy you get per kg is fairly predictable. The one yellow dot you see at the top comes from a battery pack that is assembled in both parallel and series, which boosts the energy you get per mass at the expense of a lower voltage for the same mass.
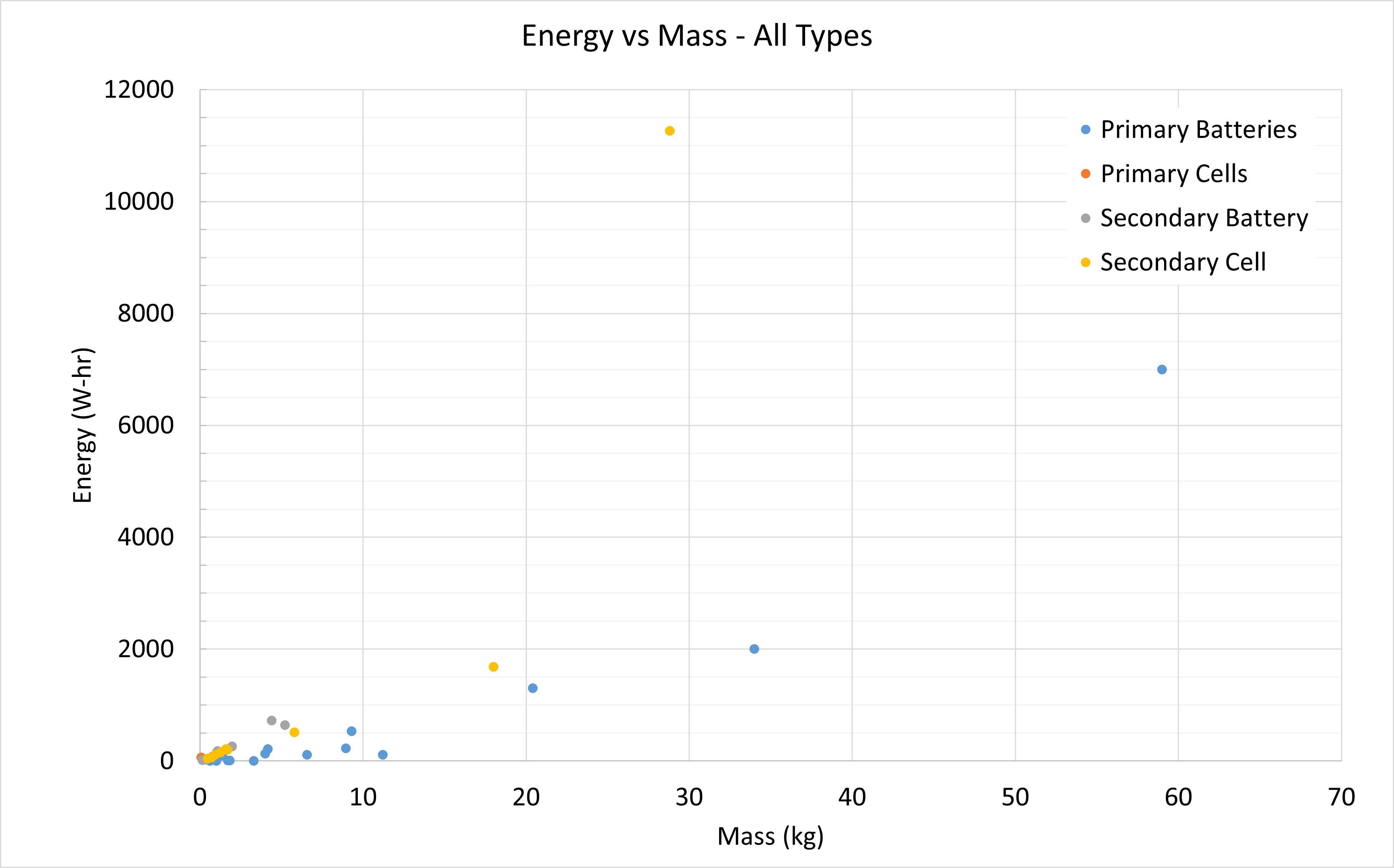
This is the same plot but zoomed in to the lower values.

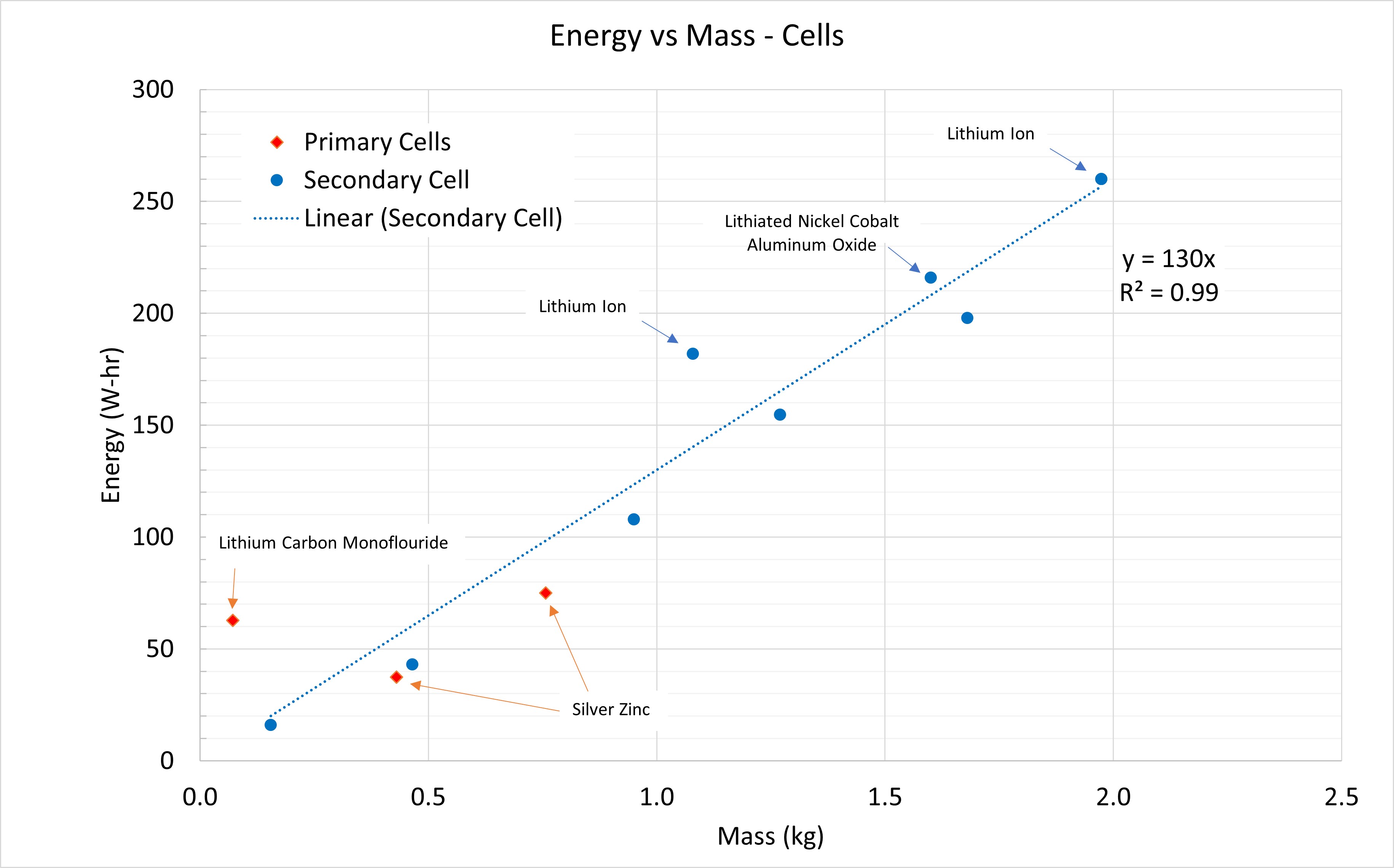
Energy vs Mass - Cell
If we look at just cells, we remove the ability to do things in parallel and series which gives us a cleaner look at trends. In this plot, we can see that regardless of battery chemistry, there’s a linear trend line. Variations within the same technology (e.g. Lithium Ion) are due to variances in vendors and cell size.
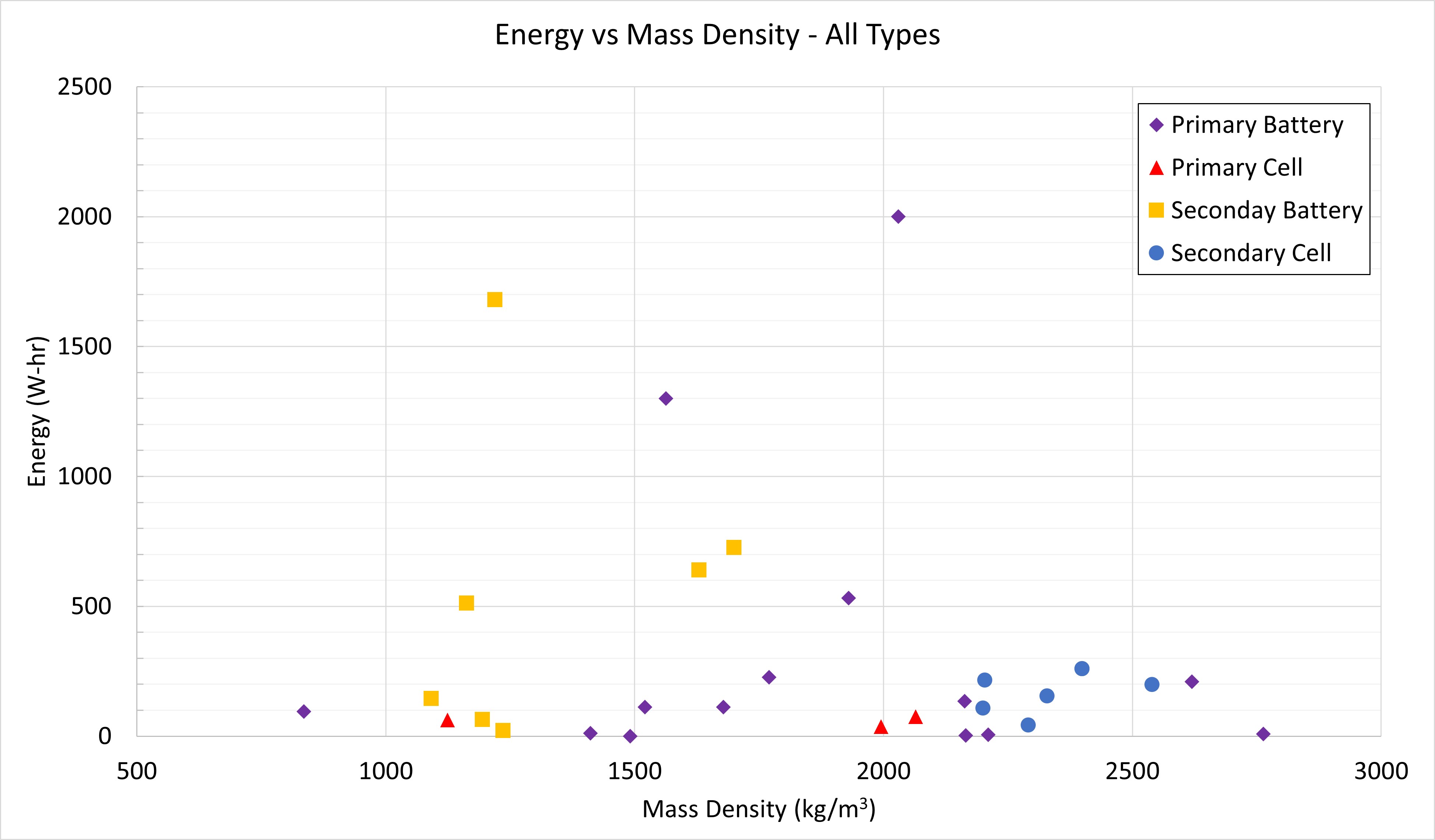
Energy vs Mass Density
The previous plots looked at mass. This one folds in volume by plotting mass density. The mass density of water is about 1,000 kg/m3 so batteries tend to be roughly 1x to 2.5x the density of water for a few hundred Watt-hours of energy. If you’re designing a CubeSat to run on batteries, you don’t have much mass available so your batteries tend to store a few Watt-hours.
Final Thoughts
Battery technology for spacecraft continues to improve. Of the vendors searched, few of them were advertising Nickel-based batteries, for instance, even though that chemistry dominated the industry not that long ago.
Another trend is the move away from primary batteries, especially for small spacecraft that can’t afford the dead weight once the batteries are drained.
Going forward, there’s some talk about nuclear-diamond batteries for low voltage applications. And governments are starting to sponsor more options for nuclear-based systems, too. Power continues to be a limiting resource on many spacecraft so it will be interesting to see what new solutions people come up with.
Curated Videos
- https://www.youtube.com/watch?v=ygwQQ13-RYo
The video editing is a bit choppy but there are plots and tidbits of information here that might make it worth your time. - https://www.youtube.com/watch?v=LqgP16JQ24I
Although not about space, it does cover different types of batteries and why you might choose them. I also like that they go through a bit of the math on calculating densities. - https://www.youtube.com/watch?v=jeisJvbASIg
Silver Zinc batteries are often used as primary batteries but not secondary batteries. This video explains why and gives a bit more insight into battery concepts, too.
Curated Links
- https://www.nasa.gov/smallsat-institute/sst-soa/power#3.4
NASA's "State of the Art Small Spacecraft Technology" pages are must-reads on many topics, including batteries. This link will take you halfway down the page to where the battery-specific section begins. - https://batteryuniversity.com/
This is an awesome, super informative website that’s well worth a read if you’re interested in learning more about battery technology. - https://www.powermag.com/battery-technology-used-in-outer-space-could-be-a-gamechanger-on-earth/
This talks about Nickel-based vs. Lithium-based batteries for space. I don’t think their comparisons to Lithium are exactly right since there are packs that can last for tens of thousands of cycles but the arguments they present are generally in line with traditional stances on batteries used in space.
 Space Steps
Space Steps